Supporting overseas
biotech companies to
enter the Japanese
market
Supporting overseas biotech companies to enter the Japanese market
We provide a One Stop Solution (consistent support from start-up to sales and licensing) to overseas/Chinese biotech companies aiming to enter the Japanese market.

We support overseas biotech companies aiming to enter the Japanese market.
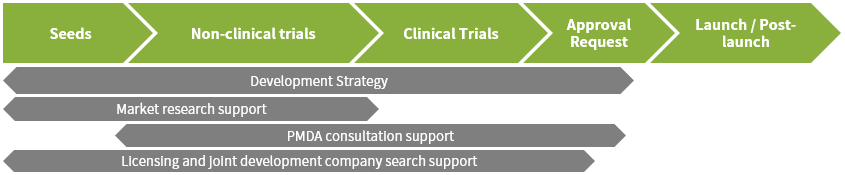
Pharmaceutical Strategy Support
- Development strategy planning
- Data Gap Analysis
- PMDA consultation support
・Participation in PMDA pre-interviews, PMDA face-to-face consultations, interpretation services
・PMDA applications and application documents, preparation of documents to be submitted, handling of inquiries - Drafting clinical trial protocols and informed consent documents
- Rare disease designation registration
Market Research
- Investigation of competing product application status/clinical development trial status
- Prediction of the number of patients with the target disease
- Survey on expected drug prices, risk assessment on drug prices
Consultation Services
- Support for development strategies
- Support for searching for potential license-out and joint development companies and setting up interviews
Strengths of EP Bio Partners Co., Ltd.
We provide a One Stop Solution (consistent support from start-up to sales and licensing out).
We provide development support mainly from experts in various fields with careers in pharmaceutical companies and bioventures.
This will facilitate good communication and ensure smooth project management.
Our staff, who speak Japanese, Chinese and English, will manage the entire study and act as a liaison between Japanese customers and Chinese operations.
Network with KOLs
We utilize our network with KOLs to conduct clinical trials while incorporating opinions from a clinical perspective.
SMO Network
Our group company, EP-Link, is the largest SMO in Japan and can help you select facilities that will encourage entry.
