About Us
EP-Bio Partners is a company that
provides overall business
planning
support for Japanese bio ventures,
academia and overseas biotech
companies that consider to
enter into Japanese Pharma Market.
We also provide development planning
and clinical trial support
for
each pipeline of Japanese bio ventures,
academia and
overseas
biotech companies.
Our Services
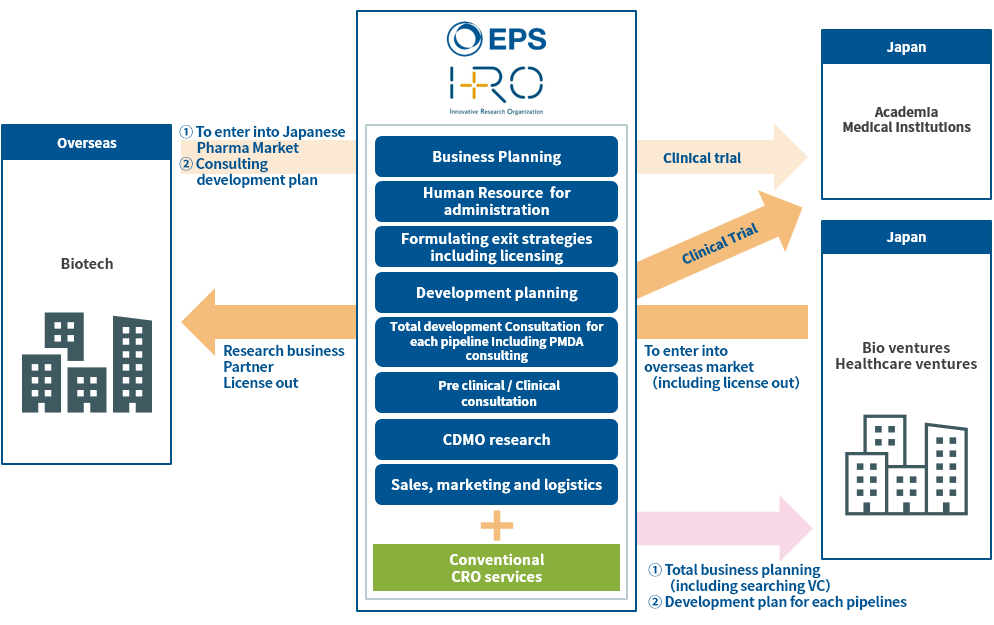
We are an EPS Group company and our main three business categories are as follows ;
1.To support developing business plans of Japanese bio ventures as well as to support development plan and clinical operation for each pipeline.
2.To support developing business plans of overseas biotech companies which plan to enter into Japanese Pharma Market, as well as to support for development plan and clinical operation for each pipeline.
3.To support Japanese pharmaceutical companies to conduct clinical trials in overseas (especially in China and other Asian countries).
Particularly, we call the above 1 and 2 business categories as an Innovative Research Organization (IRO) business which offer not only standard CRO business but also providing total solution beyond standard CRO business. Through this new business model, we aim to build a new drug discovery ecosystem originating in Japan.
Representative Director Message

Representative Director
Kosuke Kuronuma
EP-Bio Partners is a new company of the EPS Group established in June 2022 and from October 2024, it has become a subsidiary company wholly owned by EPS Holdings. Our business is an IRO* that provides development of business plans of biotech ventures and overseas biotech companies and also provide development support for each pipeline.
The business environment for drug development in Japan and overseas has changed significantly in recent years.
First of all, number of clinical trials conducted in Japan are on the downward trend. On the other hand, many biotech ventures have been establishing in Japan. In addition, there is an increase in the number of new modality drugs and the development of new drugs for rare diseases. In order to accelerate the development of these Japan-originated pipelines, several measures including financial support implemented by Japanese government for bio-ventures and academia have been increasing.
In addition, many overseas biotech companies plan to enter into Japanese Pharma Market and conduct clinical trials in Japan to obtain IND approval. In fact, the number of pipelines and clinical trials by overseas companies in Asia, China, EU and US is growing.
On the other hand, only the financial support to these companies is not sufficient to accelerate these companies’ business in Japan and development of their pipelines.
Traditionally, big pharmaceutical companies have all functions for the development inside their companies. But, to accelerate the development of Japanese bio venture companies and overseas biotech companies, a horizontal functions (VC, academia, CDMO, CMO, CRO, SMO, etc.) is necessary because these companies are not able to have all functions inside. Furthermore, in order to promote such a new system, we believe it is necessary to have a function and a role to connect, coordinate, and promote the entire process and functions to these companies.
For the overseas biotech companies, in order to start development of their pipelines there are several barriers, not only language barriers, but also some unique barriers of Japanese regulatory sciences, clinical trial systems at medical institutions and academia.
In order to solve these issues, our company’s role is to provide a comprehensive consultation service and to facilitate to help biotech ventures and overseas biotech companies smoothly for smooth development in Japan.
To achieve them, we go beyond the framework of a conventional CRO to provide one-stop solutions as an IRO, starting with consulting and leading to clinical trials and regulatory approval.
We would like to contribute to the sustainable development of the Japanese pharmaceutical industry and to the construction of a new ecosystem that includes biotech ventures, academia, and overseas biotech companies.
* IRO: Innovative Research Organization. It is not limited to conventional CRO services, but also provides support for the client’s business itself, from development planning consulting for each pipeline to clinical development.
In other words, IRO is one-stop solution service from business planning to clinical development consultation and clinical development operation.
Directors and Management
Board Member
Executive Officer Structure

Kosuke Kuronuma
Representative Director,President and Representative Executive Officer
He joined a medical startup in 2000 after experiencing sales of medical equipment and electronics equipment in Europe, North America, Asia, and Japan, and investment and financing for an electronic equipment startup in Japan for about 10 years. At that time, the company succeeded in breakthroughs that allowed e-marketing to take root in MR activities, which were mainly visits, and contributed to the company’s listing on the TSE Mothers (at the time) in 2007. He joined EPS Corporation in 2013. The company established the first subsidiary in the CRO industry specializing in clinical research and clinical trials led by physicians, enabling it to conclude contracts with almost all academia in Japan and grow into a business with a size of several billion yen. He has been a member of EPS Medical Consultancy (Japan) Co., Ltd. since its inception and served as Representative Director, President and Chief Executive Officer.

Xia Xiangming
Auditor
He started to study at Department of Genetics (now the Department of Molecular Genetics) at Okayama University as a Chinese government-sponsored student in In 1981, and afterward received a Master of Science degree. After returning to China, he spent nearly 20 years working on trade, investment, and technical exchanges between Japan and China at China Council for the Promotion of International Trade (CCPIT). He retired from CCPIT with the last position of the representative officer to Japan. In 2008, he joined EPS China Co., Ltd., EPS’s regional headquarters in China, where he was involved in the establishment and business development of EPS China, and served as President and subsequently Vice Chairman. In 2018, he was appointed Corporate Officer and Counselor of EPS Holdings, where he was primarily responsible for the management support and was the head of the managing division of EPS Creative Health Technology. He has experience in social activities, such as Vice Chairman of the Association of Chinese Enterprises in Japan and Managing Director of Chinese Chamber of Commerce Japan.

Tomohisa Hayakawa
Advisor
He was engaged in clinical development planning and project management in Japan, the U.S. and Europe at Taiho Pharmaceutical Co., Ltd. and Daiichi Pharmaceutical Co., Ltd. He joined the EPS Group in 2007 and served as the head of project management and business development in Japan, China, and Asia. On 2021, he was appointed Corporate Officer of EPS Innovative Medicine (Japan) Limited. In the same year, he was appointed Representative Director and President of EPD Co., Ltd. He has extensive experience in oncology area. He has experience in public activities such as a member of the Research Evaluation Committee of the New Energy and Industrial Technology Development Organization (NEDO) and a program committee member of the DIA Annual Conference in Japan for Asian New Drug Development. Ph. D in pharmacy and a pharmacist.

Hisayo Morioka
Executive Officer
After working as a pharmacist at a university hospital, she moved to SMO company and then joined EPS Corporation in 2004. She was mainly involved in monitoring and project management for investigator-initiated clinical trials and clinical research She later became Deputy Head of the Clinical Research Promotion Center at EP-CRSU Co., Ltd., one of EPS’s group companies, and subsequently Deputy Head of the Center of the Pharmacovigilance Center at EPS Corporation. In October 2024, she was appointed as the head of the Department of Total Solution for Overseas Companies and Executive Officer at EP-Bio Partners Co., Ltd.

Yuzo Yamamoto
Corporate Officer
After working as General Manager of the Data Science Division at EPS Corporation, he served as General Manager of the Human Resources Department of the company, and as Corporate Officer at E-Trial Co., Ltd. and EP-SOGO Co., Ltd., and as an outside director of EPS Yamanashi Co., Ltd. and EP-Mate Co., Ltd. At EPS Holdings, Inc. he served as General Manager of Planning and Promotion Department 1, promoting the improvement of the current business of the entire EPS Group, and WAY Promotion Manager, promoting the dissemination of EPSWAY, the management guideline of the EPS Group. Since April 2022, he has been responsible for promoting the Japan-China CRO business as a part-time Corporate Officer of EPS China Co., Ltd. and Corporate Officer of EPS Medical Consultancy (Japan) Co., Ltd.

Kotaro Nishio
Executive Officer
He has experience in sales of staffing & recruitment consulting service provider for R&D and clinical development, recruitment and business development at a CRO, and quality control in clinical development service at a CRO. At EPS Group, he serves as the head of the sales department for the CRO business specialized in clinical studies and investigator-initiated clinical trials, and leads the business supporting clinical studies planned by doctors, pharmaceutical companies, and medical device companies in Japan. At EPS Medical Consultancy (Japan) Co., Ltd. he is responsible for creating and promoting IRO services in Japan. EPS Innovative Medicine (Japan) Co., Ltd.

Mr. Gao Feng
Board Member
He obtained a BSc degree from Peking University, and has extensive experience in corporate planning and management for companies in China. Other than considerable experience in corporate planning and management in China, he has held senior management positions in several Chinese multinational technology companies.
Prior to 2009, he served as vice president of Beijing Securities in Hong Kong, an expert member of the National Network Culture Standardization Technical Committee of China’s Ministry of Culture, and deputy researcher at the National Cultural Industry Innovation Research Base of Peking University’s Institute of Cultural Industries, where he participated in government-led industrial projects. In 2010, he was a senior consultant in Beijing Yu Sheng Tang, contributing to development of the company’s anti-influenza specialty Chinese herbal medicine “Jinhua Qinggan” and listing the company on the Hong Kong Stock Exchange . From 2011 to 2012, he was a senior consultant for Xinhua News Agency, supporting the company’s listing on the Hong Kong Stock Exchange. From 2014 to 2017, he served as Managing Director, Chairman of the Board and Senior Consultant at Yingsheng Science Co., Ltd. and was a major shareholder during the period. He participated in the establishment of Jianhao Fund Management Company in 2018, oversaw investing in Japanese companies and dedicated to listing of the company on the Hong Kong Stock Exchange.

Takaaki Tonouchi
Executive Officer
At a domestic auditing firm, engaged in accounting audits for publicly listed companies in Japan and worked with a wide range of enterprises.At a consulting company affiliated with an auditing firm, supported the formulation of corporate restructuring plans and prepared business plans from an investor’s perspective.Subsequently, served in the corporate management department of a listed company, handling IR operations and management tasks.Joined the EPS Group in 2016, where I was responsible for budget control for group companies, building group management frameworks at EPS Holdings, and managing meetings.Since October 2025, appointed as Head of Administration and Executive Officer at EP-Bio Partners Co., Ltd.

Mao Ruifeng
Executive Officer
After working as a medical IT systems engineer, he joined Yixin China in 2012. Following his work in data science, he served as Deputy General Manager at Suzhou EPS Tigermed (Suzhou).In 2019, he transferred to EP-Techno, where he managed numerous projects as Project Manager and General Manager of Project Management.Since October 2024, he has led Japan–China development initiatives as Director of the Japan–China Department and Deputy Director of the Business Development Department at EP-Bio Partners Co., Ltd. He was appointed Executive Officer in October 2025


